Implications for life insurers

Glucagon-like peptide-1 (GLP-1) agonists are a class of drugs used to lower blood sugar and promote weight loss by activating GLP-1 receptors. Initially approved for type 2 diabetes, they are now also FDA-approved for weight management, cardiovascular risk reduction, obstructive sleep apnea, and metabolic-associated liver disease (steatohepatitis). Studies have also shown promise for other metabolic diseases, including polycystic ovary syndrome (PCOS) and obesity-related cancers.1
The most well-known GLP-1 agonist is semaglutide, sold under the brand names Ozempic (for type 2 diabetes) and Wegovy (for adults with obesity). Although this class of drugs has been FDA-approved and used to treat type 2 diabetes since 2005, their use as a treatment for obesity – and the explosion in public awareness that followed – has been a more recent development.
Due to their potential to improve mortality and morbidity outcomes, GLP-1 drugs are becoming an increasingly salient topic for insurers, particularly in relation to mortality improvement, medical underwriting, and new product development. Munich Re has previously published on the topic, including in our most recent Life Sciences Report, where we examined how these drugs may positively impact numerous drivers of obesity-related mortality, with an added scenario predicting a positive mortality effect in those with both moderate and severe obesity within insured populations.
Through a collaborative partnership between its medical and data science teams, Munich Re Life US has undertaken a large-scale study to further understand the potential significance of GLP-1 drugs. In our study, we examine:
- Trends in GLP-1 prevalence, adherence data, and potential obstacles to continued use
- Impacts on mortality among individuals taking these drugs
- Implications for assessing metabolic risk and the crucial role electronic health records (EHRs) will play
Key results
- GLP-1s may drive 0.2%–0.5% annual mortality improvement, realized over a 20-year period.
- Low adherence to GLP-1 drugs could limit long-term mortality benefits. EHRs will be vital for accurately assessing metabolic risk in underwriting.
- The mortality benefits identified for GLP-1 drugs present opportunities for carriers to transform how they assess metabolic risk in underwriting programs. However, variation in individual risk profiles, driven by poor adherence and inconsistent behavioral change, as well as other factors, underscores the need for nuance in program design and application.
Background
For our study, we leveraged a de-identified dataset representing the general U.S. population, with selected medical and prescription information appended. The dataset included information on 41 million lives, with prescription and mortality data spanning from 2015 to January 1, 2025. Ages ranged from 18 to 80, with an average age of 54, a median income of $83,000 per year, and a male-to-female ratio of 46:54.
For much of our analysis, we compared trends between diabetic and non-diabetic subpopulations. The diabetic subpopulation consists of individuals with a documented history of diabetes mellitus (excluding prediabetes), based on diagnosis codes available from 2015 onward.
The non-diabetic subpopulation includes individuals without a documented history of diabetes, as determined by diagnosis codes. GLP-1 use in this group is presumed to be for the treatment of obesity.
Prescription rates
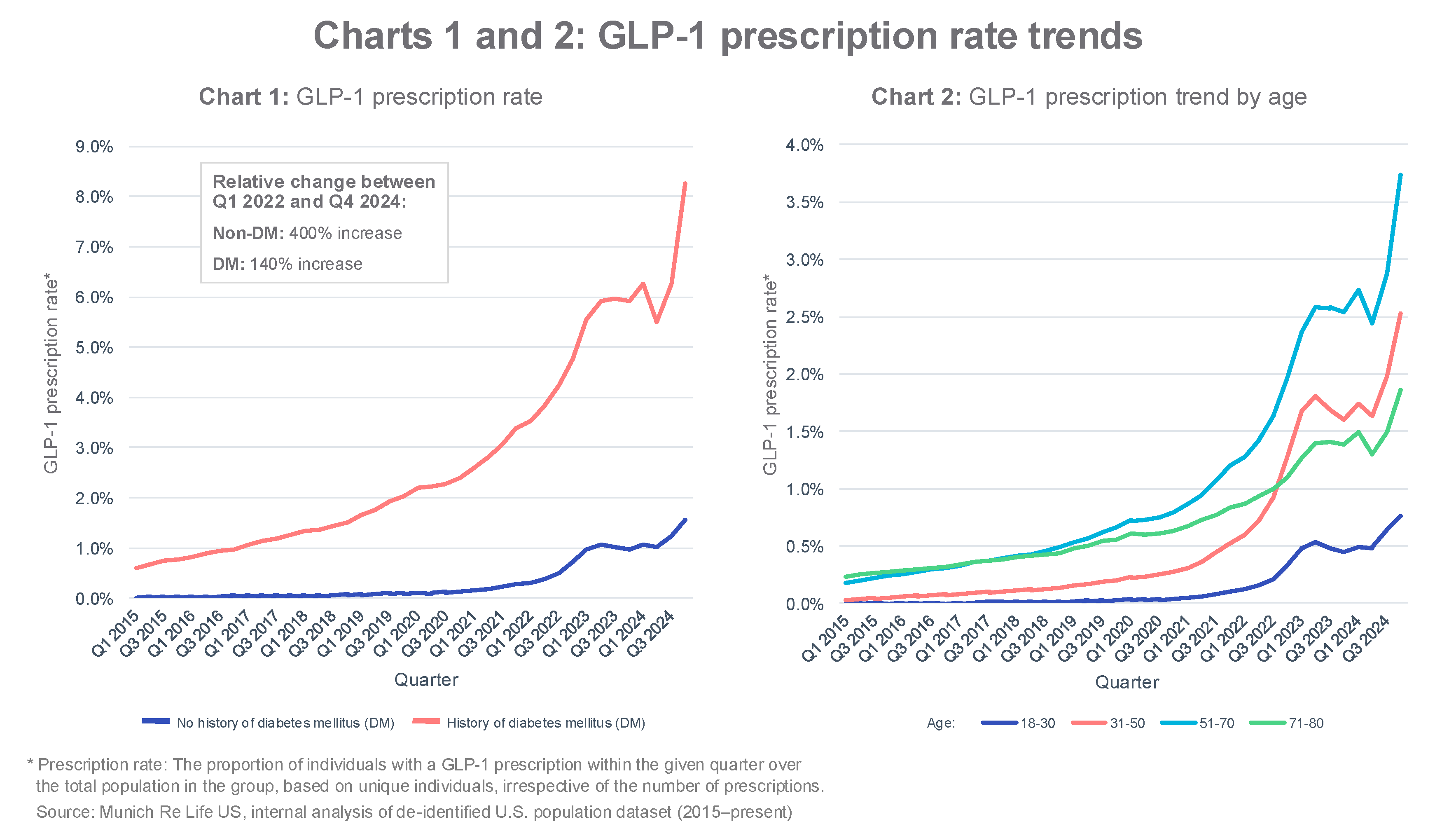
When analyzing prescription rate trends by age (Chart 2), GLP-1 drugs remain most prevalent among individuals aged 51 to 70. However, we are seeing a high growth rate among those aged 31 to 50, particularly since 2022 onward. We believe this reflects the increased proportion of GLP-1 prescriptions among younger non-diabetic individuals.
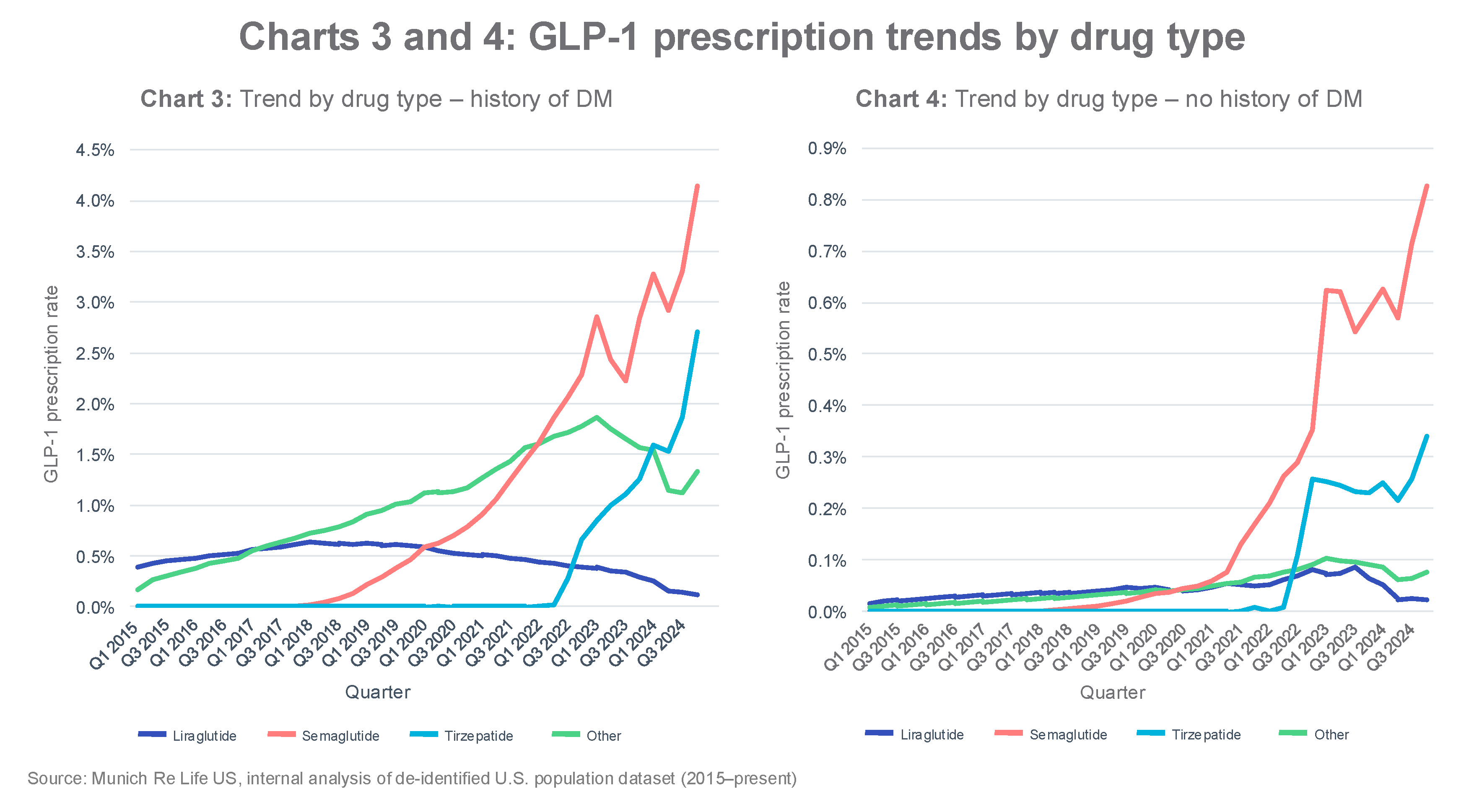
Across GLP-1 subtypes, we observe that much of the increase in prescriptions is driven by semaglutide and tirzepatide, which aligns with the dates of their FDA approvals (Charts 3 and 4).
- Semaglutide (Ozempic) was approved in December 2017 for adults with type 2 diabetes, with the brand name Wegovy approved in 2021 for chronic weight management.
- Tirzepatide (Mounjaro) was approved in May 2022 for adults with type 2 diabetes, with the brand name Zepbound approved in November 2023 for obesity.
Adherence rates: a potential obstacle?
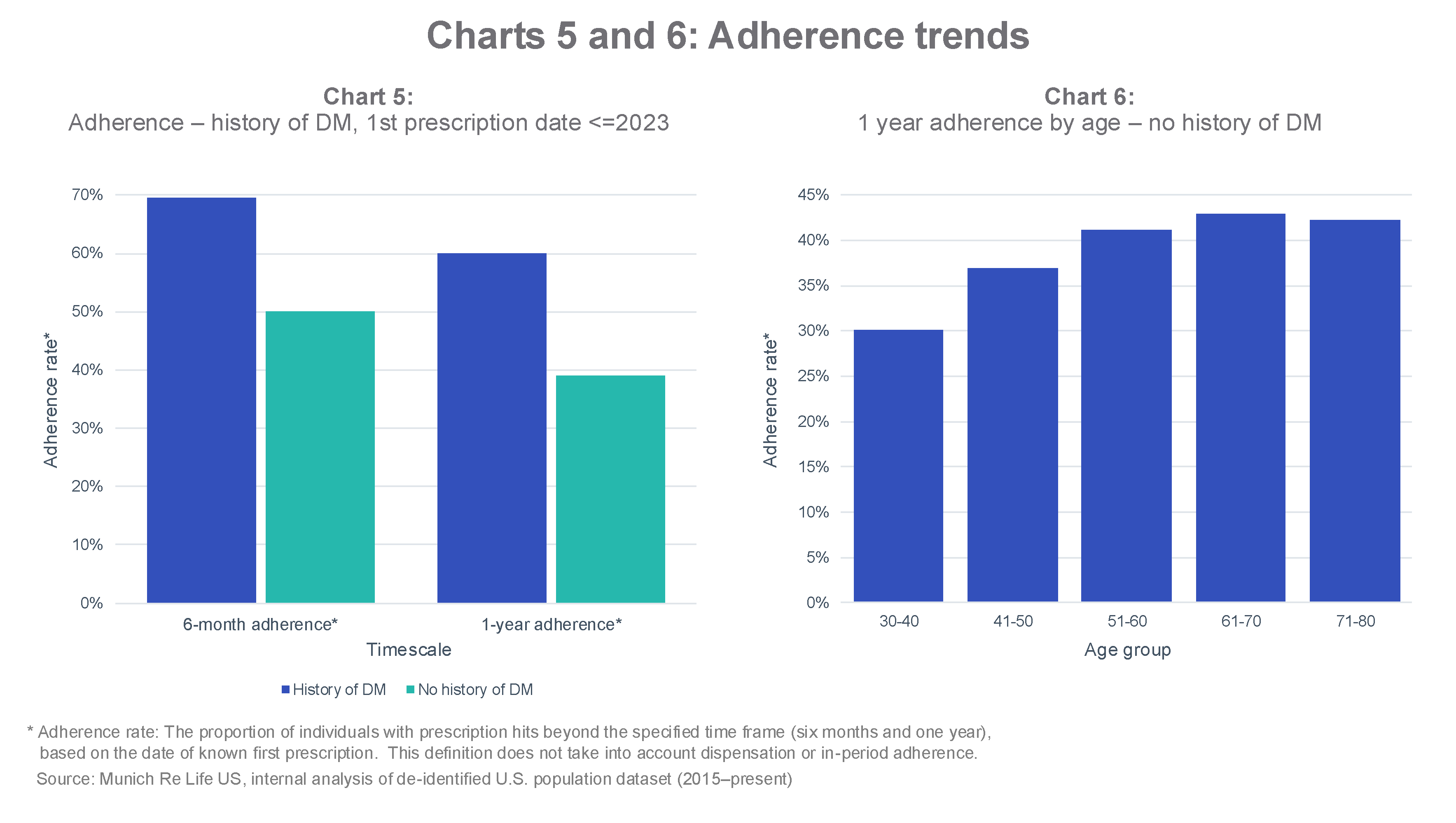
Age also plays a role in adherence, with lower rates observed in younger populations (Chart 6). Among individuals without a history of diabetes, adherence is even lower at younger ages; fewer than 30% of those aged 30 to 40 continue taking the drug beyond one year.
Numerous factors may contribute to low and inconsistent medication adherence. Adverse side effects, high medication costs, and lifestyle considerations may all play a role. For individuals taking these drugs to treat obesity, achieving weight loss targets may lead to intentional discontinuation. In some cases, this could result in rebound weight gain, especially if behavioral changes have not occurred alongside medication use.
Quantifying the mortality benefits of GLP-1s

Our analyses demonstrated favorable mortality impacts across multiple variables:
- Both diabetic and non-diabetic GLP-1 users had lower relative all-cause mortality over the study period, with very similar reductions observed in both groups compared to non-GLP-1 users.
- Lower relative mortality was consistently observed across all age groups under 60, with individuals over 60 showing less pronounced reductions.
- The favorable results were consistent across income bands, including moderate to affluent income levels, which may be more representative of insured populations.
We then incorporated the results from this exercise into the approach outlined in the Munich Re 2025 Life Sciences Report to develop a more refined estimate of future mortality improvement. Using the same assumptions as the Life Sciences Report, we forecast that existing future mortality improvement expectations could be supported by the impact of obesity drugs. GLP-1s may drive 0.2%–0.5% annual mortality improvement, realized over a 20-year period.
The way forward for understanding metabolic risk
As noted in our Life Science Report, newer obesity medications have shown unprecedented weight loss outcomes, offering the potential to reduce obesity-related mortality and morbidity risk. Our large-scale study provides additional evidence supporting a favorable mortality impact from this class of drugs. Existing research has already shown a lower risk of cardiovascular mortality related to obesity.3 Multiple large-scale cohort studies have shown a cancer risk reduction with GLP1 use in patients with type 2 diabetes and obesity.4 In addition, ongoing studies show promise that GLP-1 drugs may improve outcomes for psychological impairments, addictions, Alzheimer's disease, and Parkinson's disease.
Insurers must begin to consider the impact these medications may have on future mortality, as the effect may be substantial.
Underwriting programs: The need to reshape assessments and the fundamental role played by EHRs
Emerging evidence on the mortality impacts of GLP-1 drugs for both diabetics and non-diabetics presents opportunities to incorporate this data into underwriting risk assessments. Failing to account for GLP-1s – or including them without considering their nuanced benefit-risk profile – could place underwriting programs at a competitive disadvantage.
- Overestimation of risk: Programs may overestimate the risk in individuals experiencing lower mortality due to these treatments, leading to less competitive offers and a potential worsening of the metabolic risk pool.
- Underestimation of risk: Conversely, they may underestimate the risk in some individuals taking GLP-1s, where varied responses to treatment may lead to diverging outcomes.
- Loss of profitable business: The benefits to underwriting programs extend beyond improved ratings for those currently qualifying at substandard rates. These effects may also expand insurability for individuals currently viewed as uninsurable. Programs that ignore the impact of GLP-1s risk missing out on growth opportunities in this area.
A major challenge in using these drugs for risk assessment is obtaining comprehensive metabolic data to understand the factors influencing treatment outcomes. Among available data sources, EHRs stand out for their range, longitudinal depth, and continuity. EHRs offer the most comprehensive view of an individual’s health history, including lab results, prescription histories, and follow-up patterns. This enables insurers to gain a more accurate and dynamic understanding of metabolic health, treatment adherence, and associated behavioral risk factors.
Carriers seeking guidance on incorporating GLP-1 drugs into their underwriting framework are encouraged to contact Munich Re for support, particularly in leveraging comprehensive sources of evidence such as EHRs, where we can provide automated review and decisioning through solutions like alitheia.
Final thoughts
We are at a watershed moment in the way medical providers treat overall metabolic health. The GLP-1 class of drugs adds a groundbreaking tool in treating obesity, and a means by which we can begin reversing current adverse upward trends. Mortality gains from these drugs could change how we view metabolic risks in underwriting and unlock new opportunities to design and offer products centered around different metabolic risk profiles. Similarly, morbidity reduction from lower incidence of heart attacks, strokes, and obesity-related cancers could have parallel implications for critical illness, disability, or long-term care lines of business.
Adherence may continue to present a challenge, particularly in younger age groups; thus, insurers need to take measures to monitor and bolster adherence whenever possible to realize the best long-term mortality and morbidity benefits of these drugs.
As GLP-1s continue to reshape the landscape of obesity treatment and influence trends in long-term mortality, the ability to accurately assess metabolic risk has never been more critical. Carriers that choose to ignore this shifting landscape in their underwriting programs stand to lose out and may put themselves at a competitive disadvantage. Intelligent program design, making use of EHRs, offers a transformative opportunity to integrate robust longitudinal clinical data into risk assessment.
If you're exploring ways to integrate GLP-1s into your underwriting programs, Munich Re can provide assistance through evidence-based approaches to risk assessment, including automated EHR review and decisioning capabilities.
References
Contact the authors

/Gina%20Guzman.jpg/_jcr_content/renditions/original./Gina%20Guzman.jpg)
Related Solutions
properties.trackTitle
properties.trackSubtitle


